:max_bytes(150000):strip_icc()/chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png) 
Please report any accidental mistake in the above statistics on chemical elements. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.Ĭlick here: for a schematic overview of the periodic table of elements in chart form Although ionic radius and atomic radius do not mean exactly the same thing, the trend applies to the. Ionic radius decreases as you move across the periodic table, from left to right. In general: Ionic radius increases as you move from top to bottom on the periodic table. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The ionic radius of the elements exhibits trends in the periodic table. The unit from the Vanderwaals Radius is Pauling. The first chemical element is Hydrogen and the last is Cesium. Periodic Table - Ptable - Properties ), or, order number 1 H Hydrogen 1. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Vanderwaals Radius. Suggestions as to how the scope and content of the database can be extended are gratefully received. This list contains the 118 elements of chemistry. The following web interface allows listing and comparison of ionic and crystal radii with different coordination and charge states. The atomic radii of group 1 elemetns of the periodic table are as follows: Na(186pm),Li(152pm),R(244pm),Cs(262pm) and K(231pm). Phone: +31 152 610 900 elements listed by Vanderwaals Radius The elements of the periodic table sorted by Vanderwaals RadiusĬlick on any element's name for further chemical properties, environmental data or health effects.Separation and Concentration Purification Request.Plant Inspection & Process Optimalisation.These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and. The half of inter nuclear distance between two adjacent atoms in a metallic crystal is known as crystal radius or metallic radius or most of the time as atomic. You're having a larger atomic radius depending on how you are measuring it. Elements are organised into blocks by the orbital type in which the outer electrons are found. So as you go down the periodic table, you are getting, you are getting larger. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a 3D periodic table column style. The atomic number of each element increases by one, reading from left to right. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a 3D spiral periodic table column style. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a spiral periodic table heatscape style. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a periodic table heatscape style. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a periodic table cityscape style. There is a correlation between the atomic radii as determined from these calculations and the radii of maximum charge density in the outermost shell of the atom. You should consult reference 1 for full details, but it is not light reading for most people. These values are calculated using self-consistent-field functions (reference 1). The values given here for atomic radius are calculated values using methods outlined in reference 1. From Li to F the number of protons in the atoms increases (seen. 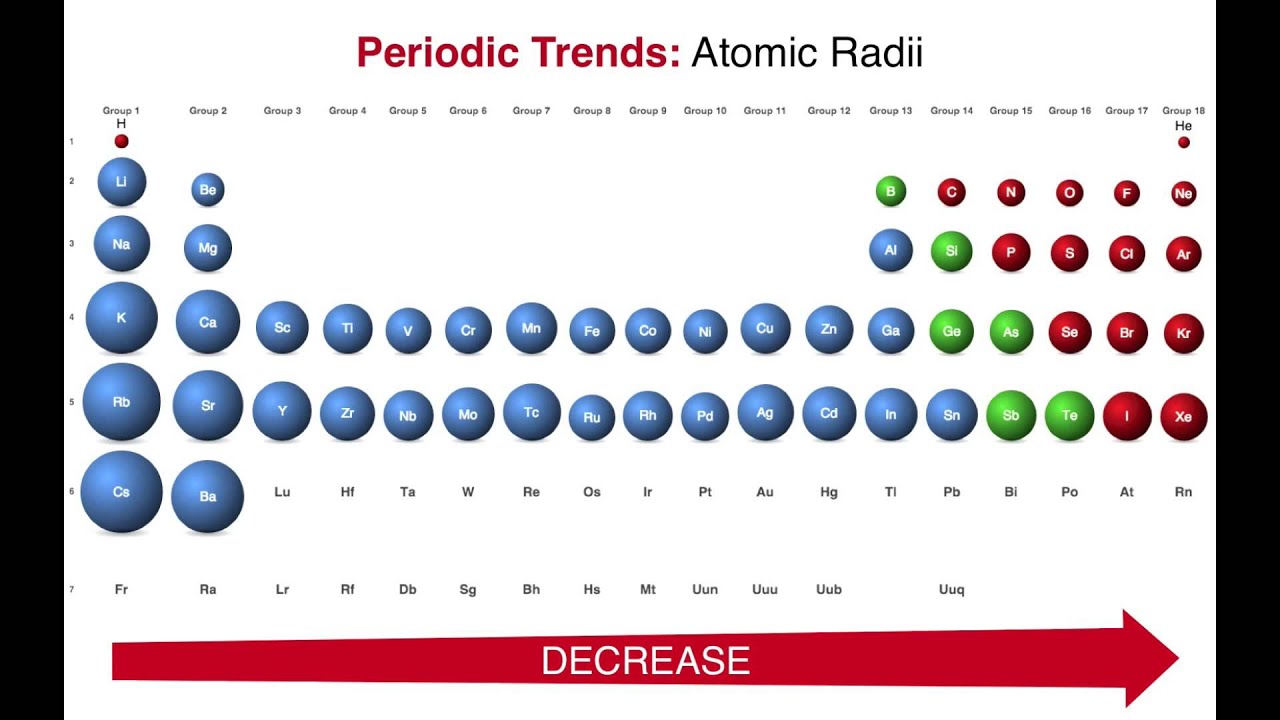 
Sometimes in text books and other sources, the rather vague term "atomic radius" is not defined and in such cases it is therefore not clear what the values actually mean. Going across the periodic table in the period of Li to F, the atomic radii of atoms decreases. Many references give table of atomic radii.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
